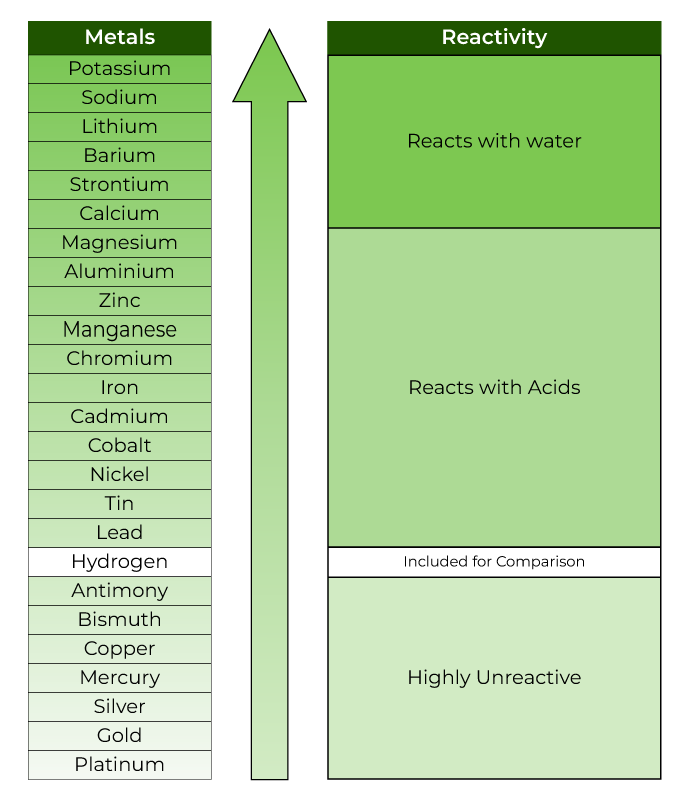
Sodium (Na): Reacts similarly to potassium, and also reacts with water to produce hydrogen gas and hydroxide ions.Ĭalcium (Ca): Reacts with water, albeit less vigorously than potassium and sodium. Potassium (K): Highly reactive metal, reacts vigorously with water. Here is the reactivity series in decreasing order of reactivity: This series helps to predict the behavior of metals when they come into contact with substances like acids or water. The reactivity series, also known as the activity series, is a list of metals and elements organized in order of their reactivity from the most reactive to the least reactive.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
